Sterile glass vial labels
Keep vials identifiable and uncompromised in sterile workflows. CleanMark sterile glass vial labels are built for small diameters, curved surfaces, and gloved handling so your processes move smoothly and your aseptic environments are protected.
Designed for traceability
From pharmaceutical fill lines to research labs, our sterile glass vial labels are pre-sterilized, double bagged, and engineered to perform reliably on curved glass without disrupting validated workflows.
Success starts with the details
Sterile glass vial labeling succeeds or fails based on how it fits into your process. We work with your team to understand every parameter that affects performance and compliance.
| Label component | Why it matters | Performance impact |
|---|---|---|
| Adhesive | Glass vials have tight curves and can behave like a LSE substrate. | Adhesives must anchor quickly and conform to tight radii to prevent lift or flagging. LSE–specific adhesive options improve initial grab and long-term adhesion. |
| Facestock | Labels often have limited surface area. | Thin, dimensionally stable synthetic materials maintain alignment and legibility on small vials. |
| Sterility processing | Labels may be applied after vials are sterilized. | Pre-sterilized labels preserve sterile status. |
| Packaging | Labels must enter sterile spaces or cleanrooms. | Double-bagging lowers contamination risk. |
| Print method | Printing often occurs in or near sterile areas | Direct thermal printing avoids ribbon handling. Thermal transfer may be used when higher durability is required. |
| Print performance | Vials require dense information in small spaces. | High-resolution, high-contrast printing keeps barcodes and identifiers readable at small sizes. |
| Temperature considerations | Vials may be filled, labeled, or stored at different temperatures. | Adhesives are selected to perform at application temperature and through expected storage conditions. |
| Process alignment | Labels must match sterility checkpoints. | Introducing a sterile label at the correct stage helps avoid contamination or unnecessary re-sterilization. |
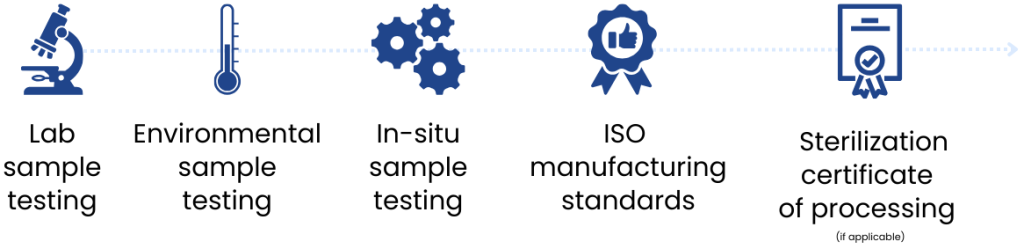
Quality control and testing
Your sterile glass vial labels go through extensive quality checks during design, sampling, and manufacturing.
Our business is to know your business
Whether you’re a semiconductor manufacturing engineer or a pharmaceutical supply chain manager, we’ll assign a team to you who is experienced in your field and knows how to solve your most common (and most complicated) challenges.
Have more questions? We have answers!
Read on for answers to our customers’ most common sterile glass vial labels questions.