Sterilization pouch labels
Protect your sterilization pouches from contamination. CleanMark sterilization pouch labels are engineered for post-sterilization application so instruments and components can be labeled after autoclave or sterilization without compromising sterile status.
Optimized for post-sterilization workflows
When autoclave and self-sealing sterilization pouches are labeled after the sterilization process is complete, the label itself must be sterile. Otherwise, the sterile barrier can be compromised. CleanMark sterilization pouch labels are pre-sterilized, double-bagged, and engineered to withstand moisture and wipe-downs.
Success starts with the details
It matters when your sterilization pouch labels are applied. When it’s after pouch sterilization, pre-sterilized labels are essential. We work with your team to understand exactly when and how your pouch labels will be applied so we can optimize materials and construction.
| Label component | Why it matters | Performance impact |
|---|---|---|
| Adhesive | Pouches may be recently sterilized or damp. | Adhesives must bond reliably to surfaces that have been exposed to steam or sterilization processes. |
| Facestock | Labels may encounter moisture or cleaning agents. | Moisture- and chemical-resistant materials maintain structural integrity and legibility. |
| Sterility processing | Labels are applied after sterilization. | Pre-sterilized labels preserve sterile status during post-autoclave application. |
| Packaging | Labels must enter sterile environments safely. | Double-bagged packaging reduces contamination risk. |
| Print method | Identification often added near sterile areas. | Direct thermal simplifies sterile workflows. Thermal transfer may be used when additional durability is needed. |
| Print performance | Pouches require clear status identification. | High-contrast, durable printing ensures sterilization dates and identifiers remain readable. |
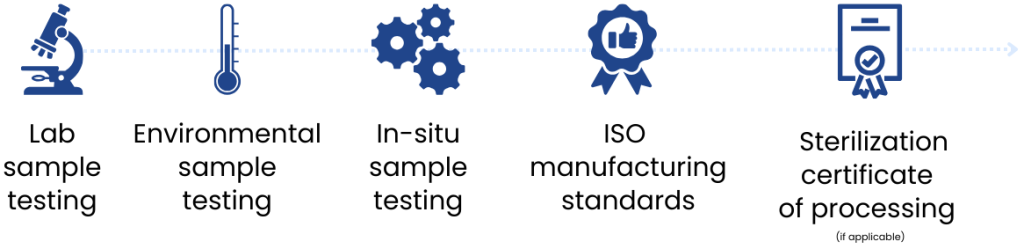
Quality control and testing
Your sterilization pouch labels go through extensive quality checks during design, sampling, and manufacturing.
Our business is to know your business
Whether you’re a semiconductor manufacturing engineer or a pharmaceutical supply chain manager, we’ll assign a team to you who is experienced in your field and knows how to solve your most common (and most complicated) challenges.
Have more questions? We have answers!
Read on for answers to our customers’ most common sterilization pouch label questions.